Research
Regulation of Microtubule Dynamics in cells
Subtle changes in microtubule dynamics critically affect all aspects of mitotic spindle function. Often small changes in microtubule assembly rates will promote chromosome instability and tumorigenesis over time.
One unusual class of kinesin-related motor proteins that we study is the Kinesin-13 Family or Microtubule Depolymerizing Kinesins. This family comprises 3 unique genes in mammals: Kif2A, Kif2B and Kif2C/MCAK or Mitotic Centromere Associated Kinesin. Our research utilizes structural and molecular mutational analysis, gene depletions and live imaging to understand the functions of these proteins in living cells, especially during cell division (mitosis). Many of the kinesin motors we study operate at the nexus between microtubule dynamics and microtubule-dependent force generation because they control microtubule length and assembly dynamics in in conjunction with their activites as motors and force generating molecules. We have found that Kif2C/MCAK may suppress microtubule assembly rates in cells in order to limit microtubule length and promote proper kinetochore attachment during mitosis. Play the video to see microtubules assembling in a live T98G cell. This activity is going on in all the cells of your body.
Behavior of Motor Molecules in vitro
In order to determine the activity that kinesin motors contribute to cells we assay their behavior on live microtubules in vitro. To the left you can see live GFP-Kif4A kinesin motors added to Alexa-568 labeled dynamic microtubules. Play the video to watch the motors and live microtubules. Most kinesin
motors use the energy of ATP to translocate along microtubules. Some classes of
kinesins will modulate microtubule assembly dynamics upon reaching the end of the
microtubule (Kinesin-4 and Kinesin-8s). Interestingly, we still do not understand what
these motors "do" to the end of the microtubule to suppress assembly. Kif18A, a
Kinesin-8 family member, will accumulate on at the plus end of kinetochore microtubules and we hypothesize that this suppresses the movement of chromosomes attached to the plus ends of micro-
tubules. The image on the right shows GFP-Kif18A in green and centromeres in red.
Chromosome Movement during Cell Division
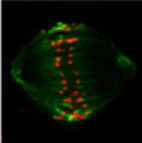
During cell division chromosomes attach to the mitotic spindle and make their way to the midpoint of the mitotic spindle where they establish a metastable position. This can be seen in the movie to the left in which centromeres (at the metaphase plate) and centrosomes at the corners are labeled
with GFP. Chromosomes oscillate in the spindle midzone until anaphase onset.
Simultaneous depletion of Kinesin motors, Kif18A and Kif4A will eliminate all the
positional cues that chromosomes use to position themselves on the mitotic
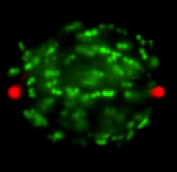
spindle resulting in chromosome that are attached to the spindle and moving but
cannot find the midzone. This is seen in the still image to the left showing a mitotic
cell in which centromeres are labeled in green and centrosomes in red.
Ironically, it is still fundamentally unknown why chromosomes need to congress
prior to anaphase. In fact, some cells that cannot congress, such as the cell on the
right, will progress into anaphase and successfully segregate their chromosomes.