![[Next]](nextbut.gif) Back to ODE Page
Back to ODE Page
To understand what makes one set of ordinary differential equations more difficult to solve than another, we need to understand how quickly each of the equations moves towards its own steady state solution.
For instance, if we have a set of ODEs that describes how the mole fraction of a component in a system and temperature of the system change with respect to time, we could try to solve them as a system of equations.
It is this fact, that one of the parameters changes quickly and the other changes slowly, that makes the set of equations stiff.
By solving for the eigenvalues for this system of equations, we can tell which
variable, y or T, is going to its steady state solutions faster.
Note.
Here is how to determine if a set of differential equations is stiff.
The two equations below are a simplified model for a catalytic
converter, where y denotes the mole fraction of CO in the gas phase and T
denotes the temperature of the gas phase. The model was simplified by excluding
the reaction term.
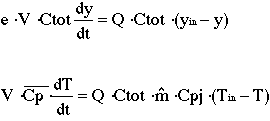
When the constants are substituted into the above equations we get: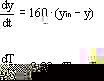

Here is the ratio of the Eigenvalues.

Page constructed by
Doug Thornhill, James Bopp, Cory Clemmons and Devin Hodgson.
For questions or suggestions, please contact us through
dt@u.washington.edu
Last modification on 7 Dec 95.