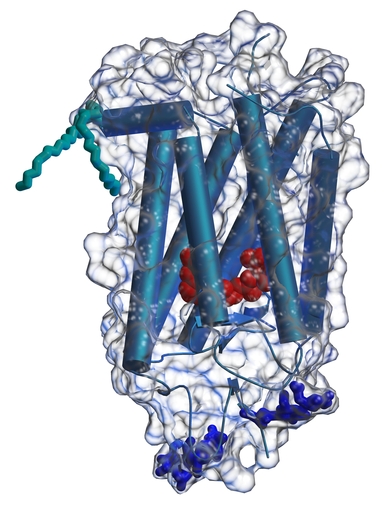

Rhodopsin is the protein found in retinas that converts light into a molecular signal that is eventually transmitted to the brain as a nerve signal. It is located in the disks in the outer segments of rod cells. When a photon is absorbed by the protein's chromophore, retinal, it undergoes a cis-trans conformational change. This structural change is carried out in the context of the protein structure and generates a change in structure on the cytoplasmic side of the protein than causes it to interact with the G-protein, transducin. A series of intermolecular interactions involving the Transducin alpha subunit and a phosphodiesterase eventually causes a reduction in the levels of c-GMP and the associated closing of ion channels in the rod cells. This causes the cells to polarize and starts the nervous signal.
We have recently solved the three-dimensional structure of rhodopsin (Science, 289, 739-745 (2000)).
Much work remains to structurally characterize the various photostates of the molecule and to determine how the structure of the surrounding protein affects the absorption properties of the chromophore in rhodopsin as well as the cone cell pigments responsible for color vision.
In addition to the relevance of this structure to vision science, it provides the first view of the 3-dimensional structure of a G-protein-coupled receptor. This family of proteins provides many drug targets for the treatment of human disease, and our structure is providing a new starting point for homology modeling and structure determinations within this group.
copyright © Ron Stenkamp stenkamp@u.washington.edu Most recent update 10/20/00
Back to Ron's homepage